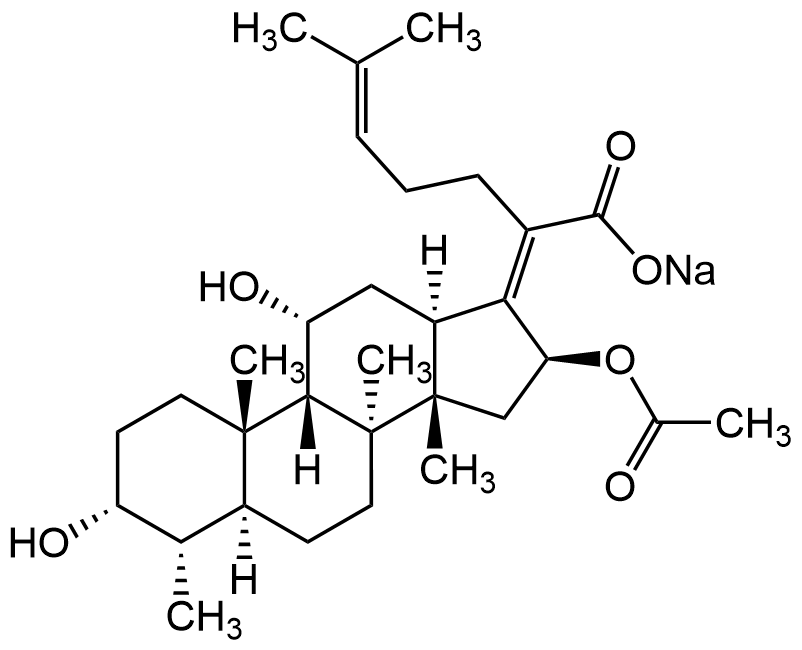
Fusidic acid sodium salt
Product Code:
CDX-F0118
CDX-F0118
Regulatory Status:
RUO
RUO
Shipping:
Ambient
Ambient
Storage:
Short term: +20°C. Long term: +4°C
Short term: +20°C. Long term: +4°C
No additional charges, what you see is what you pay! *
| Code | Size | Price |
|---|
| CDX-F0118-G001 | 1 g | £226.00 |
Quantity:
Prices exclude any Taxes / VAT
Stay in control of your spending. These prices have no additional charges to UK mainland customers, not even shipping!
* Rare exceptions are clearly labelled (only 0.14% of items!).
* Rare exceptions are clearly labelled (only 0.14% of items!).
Multibuy discounts available! Contact us to find what you can save.
This product comes from: Switzerland.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
- Further Information
- Documents
- References
- Show All
Further Information
Appearance:
White to off-white powder.
CAS:
751-94-0
Description:
The tetracyclic triterpenoid is a fusidane antibiotic originally isolated from F. coccineum. It is active against the Gram-positive bacteria S. aureus, S. pyogenes, C. diphtheriae, B. subtilis, and C. tetani but not the Gram-negative bacteria E. coli, S. typhimurium, and P. vulgaris or the fungi C. albicans and A. fumigatus. This antibacterial agent inhibits bacterial protein synthesis by preventing the release of translation elongation factor G (EF-G) from ribosomes. Fusidic acid has immunomodulatory effects and inhibits the inhibitory and activating effects of interleukins IL-1 and IL-6 on glucose-induced insulin production and exhibits antidiabetic effects in a rat model. Fusidic acid improves the symptoms of colitis in rats and inhibits the growth of Toxoplasma gondii and Listeria monocytogenes EGD in vitro, but not in mice. The sodium salt version is found to be more water-soluble.
EClass:
32160000
Form:
solid
GHS:
GHS07
Handling Advice:
Protect from light and moisture.
Hazards:
H302
InChi:
InChI=
1S/C31H48O6.Na/c1-17(2)9-8-10-20(28(35)36)26-22-15-24(34)27-29(5)13-12-23(33)18(3)21(29)11-14-30(27,6)31(22,7)16-25(26)37-19(4)32;/h9,18,21-25,27,33-34H,8,10-16H2,1-7H3,(H,35,36);/q;+1/p-1/b26-20-;/t18-,21-,22-,23+,24+,25-,27-,29-,30-,31-;/m0./s1
InChi Key:
HJHVQCXHVMGZNC-JCJNLNMISA-M
MDL:
MFCD09054714
Molecular Formula:
C31H47O6Na
Molecular Weight:
538.69
Package Type:
Vial
Precautions:
P301 + P312 + P330
Purity:
>98% (TLC)
Signal word:
Warning
SMILES:
O=C(O[Na])/C(CC/C=C(C)/C)=C1[C@@H](OC(C)=O)C[C@]2(C)[C@@]3(C)CC[C@@]4([H])[C@H](C)[C@H](O)CC[C@]4(C)[C@]3([H])[C@H](O)C[C@]2\1[H]
Solubility:
Soluble in water or DMSO.
Source / Host:
Microbial
Transportation:
Non-hazardous
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at +4°C.
Documents
References
(1) W.O. Godtfredsen, et al.; Nature 193, 987 (1962) | (2) N. Tanaka, et al.; Biochem. Biophys. Res. Commun. 30, 278 (1968) | (3) L. Verbist; J. Antimicrob. Chemother. 25, 1 (1990) | (4) K. Bendtzen, et al.; J. Endocrinol. 132, 345 (1992) | (5) K. Buschard, et al.; Autoimmunity 14, 101 (1992) | (6) J Turnidge, et al.; Int. J. Antimicrob. Agents 12, S23 (1999) | (7) P. Collignon & L. Turnidge; Int. J. Antimicrob. Agents 12, S45 (1999) | (8) K. Christiansen; Int. J. Antimicrob. Agents 12, S73 (1999) | (9) A. Savelsbergh, et al.; RNA 15, 772 (2009)