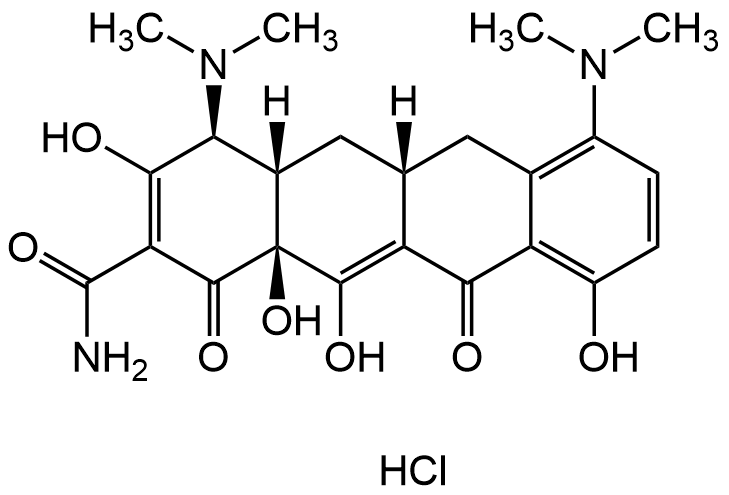
Minocycline hydrochloride
Product Code:
CDX-M0620
CDX-M0620
Regulatory Status:
RUO
RUO
Shipping:
Ambient
Ambient
Storage:
Short term: +20°C. Long term: +4°C
Short term: +20°C. Long term: +4°C
No additional charges, what you see is what you pay! *
| Code | Size | Price |
|---|
| CDX-M0620-M250 | 250 mg | £75.00 |
Quantity:
| CDX-M0620-G001 | 1 g | £151.00 |
Quantity:
Prices exclude any Taxes / VAT
Stay in control of your spending. These prices have no additional charges to UK mainland customers, not even shipping!
* Rare exceptions are clearly labelled (only 0.14% of items!).
* Rare exceptions are clearly labelled (only 0.14% of items!).
Multibuy discounts available! Contact us to find what you can save.
This product comes from: Switzerland.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
- Further Information
- Documents
- References
- Show All
Further Information
Appearance:
Yellow powder.
CAS:
13614-98-7
Description:
Minocycline, a semi-synthetic second-generation tetracycline, is a broad spectrum antibiotic with bacteriostatic function. Tetracyclines, including minocycline, function by binding to the 30S ribosomal subunit, thereby preventing charged tRNA from delivering amino acids to elongate the protein chain and form cellular protein. This disruption results in a bacteriostatic effect on the prokaryotic cell, leading to the loss of its ability to grow or replicate. Being lipid-soluble compounds, tetracyclines can traverse hydrophobic barriers such as biological membranes. It is orally active and brain penetrant. Apart from its efficacy against gram-positive and gram-negative bacteria, minocycline also exhibits antioxidant, anti-apoptotic, anti-tumor, anti-inflammatory, immunomodulatory, and neuroprotective properties. Minocycline has anti-inflammatory properties and inhibits neuroinflammation in pre-plaque of Alzheimer's disease-like amyloid pathology through inhibition of key inflammatory enzymes like inducible nitric oxide synthase (iNOS), matrix metalloproteinase 9 (MMP-9) and 5-lipoxygenase. Minocycline inhibits endothelial cell proliferation and angiogenesis, and inhibit the enzymatic activity of poly(ADP-ribose) polymerase-1 (PARP-1). Minocycline exhibits anti-tumor activity in glioma by inhibiting membrane type 1 matrix metalloproteinase (MT1-MMP). Minocycline increases cognition and neuronal differentiation.
EClass:
32160000
Form:
solid
Handling Advice:
Protect from light and moisture.
InChi:
InChI=1S/C23H27N3O7.ClH/c1-25(2)12-5-6-13(27)15-10(12)7-9-8-11-17(26(3)4)19(29)16(22(24)32)21(31)23(11,33)20(30)14(9)18(15)28;/h5-6,9,11,17,27,29-30,33H,7-8H2,1-4H3,(H2,24,32);1H/t9-,11-,17-,23-;/m0./s1
InChi Key:
GLMUAFMGXXHGLU-VQAITOIOSA-N
MDL:
MFCD00083669
Molecular Formula:
C23H27N3O7 . HCl
Molecular Weight:
493.94
Package Type:
Vial
Purity:
>98% (HPLC)
SMILES:
CN(C)[C@H](C(O)=C1C(N)=O)[C@@]2([H])[C@@](C(O)=C(C(C3=C(O)C=CC(N(C)C)=C3C4)=O)[C@]4([H])C2)(O)C1=O.Cl
Solubility:
Soluble in DMSO (30mg/ml), DMF, methanol or water (5mg/ml).
Source / Host:
Synthetic
Transportation:
Non-hazardous
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at +4°C.
Documents
References
(1) R.J. Tamargo, et al.; Cancer Res. 51, 672 (1991) | (2) S. Gilbertson-Beadling, et al.; Cancer Chemother. Pharmacol. 36, 418 (1995) | (3) M. Kloppenburg, et al.; Clin. Exp. Immunol. 102, 635 (1995) | (4) S. Parangi, et al.; PNAS 93, 2002 (1996) | (5) T. Tikka, et al.; J. Neurosci. 21, 2580 (2001) | (6) V. Brundula, et al.; Brain 125, 1297 (2002) | (7) F. Giuliani, et al.; J. Leukoc. Biol. 78, 135 (2005) | (8) M.C. Zink, et al.; JAMA 293, 2003 (2005) | (9) S.S.V. Padi & S.K. Kulkarni; Eur. J. Pharmacol. 601, 79 (2008) | (10) R. Tao, et al.; J. Cardiovasc. Pharmacol. 56, 659 (2010) | (11) N. Garrido-Mesa, et al.; Pharmacol. Res. 67, 18 (2013) | (12) N. Garrido-Mesa, et al.; Br. J. Pharmacol. 169, 337 (2013) | (13) D.J. Ritchie & A. Garavaglia-Wilson; Clin. Infect. Dis. 59, S374 (2014) | (14) O.M. Dean, et al.; Clin. Psychopharmacol. Neurosci. 12, 180 (2014) | (15) J.A. Brenes-Salazar; Can. J. Physiol. Pharmacol. 93, 863 (2015) | (16) J. Budni, et al.; Curr. Alzheimer Res. 13, 1319 (2016) | (17) A.R. Afshari, et al.; J. Oncol. 2020, 8659802 (2020) | (18) D.A. Shin, et al.; J. Pain Res. 14, 139 (2021) | (19) S. Singh, et al.; Curr. Mol. Pharmacol. 14, 1046 (2021) | (20) A. Rezaei, et al.; Naunyn Schmiedebergs Arch. Pharmacol. 397, 2835 (2024)